Sodium Bicarbonate: Old Science with New Opportunities
Historical Development
Sodium bicarbonate, familiar to most as baking soda, has traveled quite a path since the early days of industrial chemistry. The world saw its first large-scale production in the late 18th and 19th centuries, thanks to processes like the Solvay method, which replaced older, less efficient ways to obtain soda and related chemicals. Growing up in a house where kitchen chemistry was valued, I learned to appreciate the historical connection between science and common products. Sodium bicarbonate became a staple, not just because it was cheap, but because industry found ways to produce it reliably and in large amounts. The ripple effect continues today, with nearly every home and business relying on its gentle yet effective power. Historical records also show medical uses going back centuries, often touted for easing indigestion long before clinical trials became the scientific standard.
Product Overview
The white, powdery look of sodium bicarbonate doesn’t hint at the range of uses concealed in those tiny crystals. So many people have learned about it through home remedies, cleaning solutions, or baking. The real story is that this compound serves vital functions across industries. Food, medicine, fire-fighting, animal care, textiles, and water treatment—nearly every major sector uses it either plainly or as a component of blends. The product can come in varying grades, from technical to pharmaceutical, reflecting differences in purity and oversight. As a consumer and sometime hobby baker, I’ve seen firsthand how “baking soda” runs laps around specialty “magic” cleaners and expensive digestive aids. This sort of versatility points to an essential ingredient that quietly underpins routine modern life.
Physical & Chemical Properties
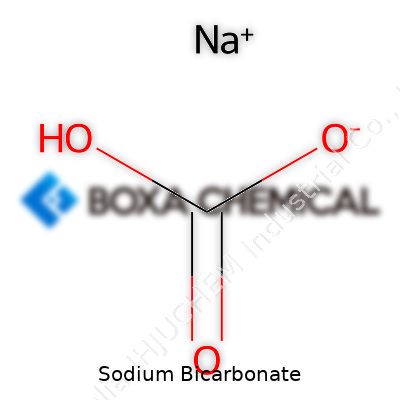
Sodium bicarbonate’s secret is its chemical resilience and mild nature. Its physical form—a fine, white crystalline powder—dissolves in water to create a slightly basic solution with a pH just above neutral. Chemically, NaHCO3 acts as a weak base and decomposes easily when heated above 80 degrees Celsius, producing carbon dioxide, water, and sodium carbonate. The fine line between stability under everyday conditions and readiness to react makes it ideal for food leavening, gentle cleaning, and rapid release tasks like fire suppression. It weighs a little more than water and refuses to burn or support flames, adding safety as a bonus feature. Years of kitchen experiments taught me that not every household powder can handle a range of temperatures and stay active, but sodium bicarbonate never disappoints.
Technical Specifications & Labeling
It’s an odd world where shoppers learn chemistry through nutrition panels or ingredient labels. Sodium bicarbonate always shows up as either “baking soda” for food or under its full name for specialty uses. Pharmaceutical and food grades undergo extra purity screening, often showing less than 0.5% impurities including sodium carbonate and chloride. Labels also reveal its role as a leavening agent (E500 in Europe) and sometimes list warnings about moisture exposure or storage recommendations—a direct callback to how quickly it absorbs water from the air. I learned to store my supply in airtight containers after too many humid summers left my kitchen tin clumpy and cakey. Industry specs drive consistent product, but the fine print also keeps consumers and workers aware of quality differences.
Preparation Method
Large-scale production depends on chemical finesse. The Solvay process stands as the reigning champion, transforming sodium chloride and ammonia with carbon dioxide under carefully controlled conditions. This recipe doesn’t just create sodium bicarbonate; it also keeps energy and waste budgets in check, which has long-term benefits for the environment and operating costs. For smaller batches or specialty needs, chemical suppliers may use alternative routes, but the core chemistry rarely changes. Home chemists can even create a tiny batch by mixing vinegar with a pure alkaline source, but don’t expect to scale up without the right equipment. Understanding the balance of raw materials and the necessity for byproduct management opened my eyes to just how much planning lives behind a humble box found at the corner grocery.
Chemical Reactions & Modifications
Sodium bicarbonate comes with a bag of tricks rooted in its ability to react with both acids and heat. Add an acid, you get a fizz of carbon dioxide and water—exactly the lift needed in cake batters and antacid tablets. Heat it, and the same gas release gives fire extinguishers their non-toxic punch. Strip out the carbon dioxide, and you’re left with sodium carbonate, another useful industrial chemical. Chemical engineers have long found ways to tweak its structure, turn it into blends, and exploit its buffering properties. My own forays into kitchen science mirrored this; learning to adjust acidity in baking made me realize just how precise—and forgiving—this chemical can be. The pathways are simple, but their impact carries forward through hundreds of applications, simplifying daily life.
Synonyms & Product Names
The same chemical wears many names, some rooted in chemistry labs, others in marketing plans. Most homes know it as baking soda, but commercial packaging may list it as bicarbonate of soda or sodium hydrogen carbonate. In medical circles, expect to find it as “sodium bicarb” during emergency treatments. The acronym E500 pops up on food labels, which can confuse folks who don’t follow food science. Trade names tend to follow branding rules, but at the end of the day, it’s the same reliable compound. Every trip to the store or scan of ingredient lists reminds me that one chemical can play many public roles, depending on who’s asking and where they look.
Safety & Operational Standards
Safety with sodium bicarbonate rarely triggers alarm bells, but regulations don’t let it slip under the radar. Health agencies measure its dust production, check for hidden contaminants, and push for smart storage practices to fend off moisture and foreign materials. Occupational standards in manufacturing call for proper containment, eye protection to guard against airborne particles, and guidelines on safe disposal of residue. In the food and pharmaceutical sectors, oversight ramps up so nobody ends up with a tainted or mislabeled product. I recall my early jobs in restaurant kitchens—kitchens thrive on sodium bicarbonate but treat it with respect, especially around acids and hot surfaces. It’s an easy chemical to handle, but responsible practice ensures it stays a helpful tool, not a source of trouble.
Application Area
No single chemical lands in so many unrelated fields. From the kitchen to the emergency room, sodium bicarbonate plays a silent supporting role. As a leavening agent, it drove the birth of “quick bread” and cleaned household surfaces before fancy formulations came along. In hospitals, clinicians turn to sodium bicarbonate as a buffer to treat conditions like metabolic acidosis, or as an antidote in some poisonings. Municipal water treatment plants rely on it to balance pH and soften water, helping stretch the lifespan of pipes. Farmers feed it to livestock to keep digestive systems working evenly. Firefighters depend on dry powder extinguishers packed with sodium bicarbonate to tackle grease or electrical fires, buying precious seconds where water would only make things worse. As a gardener, I’ve found its mild alkalinity helps tame soil acidity, showing how straightforward chemicals have far-reaching value. Across this whole array, it brings certainty and dependability to tasks that span far beyond its origins.
Research & Development
Fresh curiosity and shifting policy steer research into sodium bicarbonate’s new uses. Environmental concerns over industrial emissions have engineers testing it as a capture agent for flue gases rich in CO2. Food scientists explore how sodium bicarbonate can blend with natural acids to create “clean label” products, stripping away unnecessary artificial ingredients. Medical researchers dig into its impact on human health, from potential roles in cancer therapy to tackling chronic kidney disease. My own experience in science education makes me appreciate the spirit of innovation heavy in the studies on this old standby. Researchers don’t stop at traditional uses but treat this compound as an open book, pushing for processes that waste less energy and cut down on byproducts. These efforts offer hope that an old solution may keep pace with new global challenges.
Toxicity Research
Sodium bicarbonate falls into the rare category of chemicals judged safe for broad exposure in food, medicine, and cleaning—at the right dose. Problems arise only with overuse or in sensitive individuals. Doctors keep an eye out for sodium overload or alkalosis, especially in kidney patients. Animal studies back up its general safety, but also underscore the need for moderation. There’s little evidence of chronic toxicity outside of improper industrial handling or substantial accidental ingestion. Parents and teachers who lean on sodium bicarbonate as a classroom or household science tool get peace of mind from decades of use and testing. Proper labeling and common sense round out the picture, reminding everyone that even the mildest chemicals deserve respect.
Future Prospects
With all that’s changing in technology and society, sodium bicarbonate’s story is far from finished. Clean energy researchers are looking to sodium-based compounds as tools to capture and lock away greenhouse gases, which could help trim emissions from power plants. Food and beverage firms are under consumer pressure to avoid unfamiliar additives, which brings simple, well-studied chemicals like sodium bicarbonate into higher demand. In healthcare, research pivots toward its use as a buffer in new therapies and as a sidekick in more effective drug delivery. Shifts in industrial chemistry focus on making its production more sustainable, using renewable or recycled inputs instead of traditional mined salts. I can see sodium bicarbonate keeping its reputation for usefulness while being folded into smarter, greener approaches across the board. As the modern world looks to trusted, accessible solutions, this basic powder sits ready for whatever the future demands.
The Everyday Hero in Your Kitchen and Beyond
Baking a cake, cleaning a smelly fridge, or nursing a bout of heartburn—sodium bicarbonate shows up in places that matter in daily life. Most people know this white powder as baking soda, tucked away in kitchen cupboards across the country. Beyond its role in baking, it wears many hats, proving its value every single day.
Baking, Cleaning, and Health: Practical Uses
Pull out a cookie recipe, and there it is—baking soda. It reacts with acids like buttermilk or lemon juice, releasing carbon dioxide and lifting cakes and cookies to fluffy heights. This isn’t just kitchen science; it’s a culinary trick that’s been trusted for generations. Without this fizz, our favorite baked goods would miss their signature texture.
Cleaning takes effort and patience. Many harsh chemicals claim to wipe out stains and odors, but sodium bicarbonate does the job without strong smells or eye-watering fumes. Sprinkle some in a stinky gym bag, and the odors fade out. Coffee stains on counters fade after a gentle scrub. Mix a paste to clean ovens. I’ve even used a bit to freshen up sneakers. Safe for skin and non-toxic, it beats out complicated cleaning sprays cluttering the cabinet.
Stomach aches from spicy food or too much coffee feel miserable. Reach for a glass of water with a bit of baking soda, and relief follows soon. Sodium bicarbonate calms an upset stomach and heartburn by neutralizing excess acid. Pharmacists have offered this remedy for generations. It’s important, of course, to use only the recommended amounts—overdoing it could bring its own problems.
Underappreciated HVAC and Medical Roles
Few people think about sodium bicarbonate outside the house, but hospitals and clinics use it too. It’s added to certain medications or IV drips to correct the body’s pH during medical emergencies, especially in cases involving too much acid in the blood. Emergency rooms count on it, showing how this simple compound holds a spot in modern health care that isn’t obvious at first glance.
Fire extinguishers labeled “B” or “C” often carry sodium bicarbonate as the active ingredient. Powder smothers grease or electrical fires, a real advantage in restaurant kitchens or workshops where water would only spread the problem. It’s helped to stop more than a few emergencies before fire crews arrive.
Considerations and Moving Forward
People often forget small things can solve big problems. Sodium bicarbonate tends to be overlooked as just another kitchen item, yet it steps up in cooking, cleaning, health, and even crisis situations. Cutting back on harsh commercial cleaners and choosing sodium bicarbonate encourages a safer home while keeping costs down. In health, responsible use of such remedies can lighten the load on over-the-counter drugs.
For those chasing a greener, less wasteful lifestyle, the old box of baking soda offers solutions. Less packaging, lower toxicity, and fewer chemicals in the environment matter more and more. Tapping into knowledge that’s been passed down makes life more manageable and opens doors to simple fixes that actually work.
The Everyday Nature of Baking Soda
Baking soda, or sodium bicarbonate, might feel almost invisible in a crowded kitchen, but it fills a lot of roles. I remember being a kid and watching my grandmother dust a bit into her cookies, swearing it made them fluffier. Later, she would reach for the same box as a quick fix for the stomach aches that sometimes followed an overly ambitious meal. Decades later, some things don’t change: families still use it for baking, cleaning, and settling mild indigestion.
Science and Trust: What We Know
From a science perspective, there’s solid evidence behind those old-school remedies. Sodium bicarbonate belongs to the alkaline salt group, and it neutralizes acids. When it hits stomach acid, it produces water and carbon dioxide, creating a relieving burp and easing heartburn symptoms. That’s why antacid tablets use it as an active ingredient.
Doctors recognize this effect, and the U.S. Food and Drug Administration lists sodium bicarbonate as "generally recognized as safe" when used in food and for short-term relief of acid indigestion. So, mixing a pinch into pancakes or reaching for it when heartburn flares up falls within accepted guidelines.
Safety comes down to quantity and frequency. A normal helping in a baked good, or the occasional antacid dose, won’t trouble most folks. Health authorities confirm that the body processes the resulting compounds, and the kidneys work to restore acid-base balance.
Where the Problems Start
Dangers show up only when someone starts overusing it. I’ve known people who believed that if a little neutralizes acid, a lot must be better. That logic can backfire. Taking too much can disrupt the body’s pH, raising blood alkalinity. This puts extra stress on the kidneys, and it can even slow breathing or trigger muscle cramps. Nurses and poison control centers deal with calls about overuse more often than you’d imagine.
Those with high blood pressure, heart disease, or kidney issues face risks even with moderate use, mainly because sodium bicarbonate carries a fairly heavy sodium load. High sodium intake links to fluid retention and increased blood pressure. Anyone following a low-sodium diet would do well to check with a healthcare provider before taking sodium-based remedies, even ones that seem harmless.
Common Sense and Better Choices
Baking soda works as a reliable kitchen helper and occasional medicine, but it’s not a fix-all. Labels on antacids now remind us not to use them for more than two weeks without medical advice. If someone keeps reaching for the box to handle acid reflux, something bigger might need attention.
When in doubt, talk to a registered dietitian or a doctor. This isn’t about denying the freezer-burned wisdom of grandmothers everywhere, just about backing it up with some modern caution. Using sodium bicarbonate in moderation stays well within safe limits for almost everyone, but the key is staying mindful, reading ingredient labels, and knowing when to ask a professional.
Balance Over Band-Aids
It’s tempting to treat symptoms quickly with an easy at-home fix, but bigger changes—like cutting back on processed foods, keeping portions in check, and staying active—do more good for digestive health in the long run. Everything in its place: baking soda belongs in cookies and quick fixes, not as an everyday supplement.
Sodium Bicarbonate: More Than Just a Baking Aid
There’s a box in most kitchen cupboards that rarely draws attention: baking soda, or sodium bicarbonate. Most folks know it from recipes, but for a long time, people have used this humble powder as a cleaning agent too. My grandmother kept an open box of it in her fridge, but she also grabbed it as her go-to for stains on the counter, pans with stuck-on grime, and even to freshen up old rugs. She didn’t need high-tech sprays or fancy wipes. That little box did more than advertised, and evidence supports this kind of kitchen wisdom.
Why Sodium Bicarbonate Works for Cleaning
Sodium bicarbonate has mildly abrasive qualities. It helps lift sticky residues on stovetops or tiles without scratching most surfaces. That gritty texture comes in handy for scrubbing—a spoonful on a damp sponge will tackle dried spills or soap scum without damaging enamel or steel. Mix it with water to make a paste for the inside of the oven or stubborn marks in the bathtub. Its chemical nature helps neutralize odors, too. That’s not just folklore. Sodium bicarbonate reacts with acids and bases, breaking down molecules that cause smells or stains.
The Cost and Eco Angle
Nowadays, there’s real concern about household products and what they do to health and the environment. Cleaning sprays often pack a punch of synthetic fragrances, bleach, or ammonia. Sodium bicarbonate sticks to basics; it’s non-toxic (unless you go wild eating it) and breaks down easily. No chemical fog, no worries about what gets left in the air or washed down the drain. The cost is hard to ignore. You can grab a big box for the price of a fancy coffee, and it keeps for ages. For families watching their budgets, or anyone tired of buying a separate cleaner for every job, that holds a real appeal.
What It Can and Can’t Do
Some jobs suit it better than others. Greasy pans, stained mugs, spills in the fridge—it’s hard to beat. I’ve used it to freshen sneakers and deodorize litter boxes. No perfume, just a clean smell. On tougher tasks, like baked-on oven gunk or rust, it helps, especially if you add a dash of vinegar to spark a fizzing reaction. Just keep in mind, it won’t kill most bacteria or viruses. For that, soap and hot water or a disinfectant do better. Still, for everyday dirt, odors, or gentle polishing, it pulls more than its weight.
Potential Solutions for Cleaner Living
A lot of homes could cut back on chemical cleaners by giving sodium bicarbonate a try on basic chores. Schools could share old-fashioned cleaning tips. Local governments might suggest safer options in waste reduction campaigns—simple changes like this make a difference over time. It’s not a one-size-fits-all solution, but stocking this powder gives everyone a low-cost backup for most messes. As more people look for safer alternatives, it’s worth returning to old-school tricks that combine science with common sense.
Baking soda’s success in the cleaning aisle comes from real experience, confirmed by simple chemistry. Grab a box—chances are, it’ll keep living up to its reputation many years from now.
Looking Past the Label
Sodium bicarbonate sounds plain, but it sits in medicine cabinets and kitchens everywhere. Folks reach for it to knock out sour stomachs or heartburn, chase away odors, or even help with kidney troubles under a doctor’s watch. It looks friendly, and for the most part, it delivers on those small promises. But too much of a good thing brings its own set of problems, and sodium bicarbonate is no different. Anyone considering it—either in a glass of water or hidden in their food—should know where things can go off track.
Common Side Effects: More Than Just Gas
A little bit of sodium bicarbonate can take the edge off acid in the stomach, giving a break from burning discomfort. Take too much, though, and that fizzing relief can turn to bloating, gas, and sometimes belly cramps. In my own experience, it brings on burping—sometimes so much that it’s almost embarrassing in a quiet room. Some people pick up on nausea or a mild headache, especially if they’re not used to it.
These symptoms usually move on fast. The real trouble starts for those with underlying health issues or who end up overusing it. This common kitchen item is mostly sodium. That doesn’t raise eyebrows at first, but the body notices. If someone already has heart disease, high blood pressure, or problems with fluid retention, the extra sodium can ramp those struggles up. More sodium means the body hangs onto more water, adding to swelling, puffiness, and stressing out the heart.
Hidden Dangers: Too Much of a Good Thing
The biggest risk I’ve seen comes from taking sodium bicarbonate often without realizing how quickly things add up. High doses can push the body’s balance of acids and bases out of whack. There's even a real risk of something called metabolic alkalosis—a fancy way of saying your blood tips toward being too alkaline. This triggers confusion, muscle twitching, and, if unchecked, serious seizures or heart rhythm problems.
There’s also the issue of interactions with other medicines. Mixing sodium bicarbonate with calcium supplements, diuretics, or certain heart medicines can shift blood chemistry in unpredictable ways. Kidneys usually keep this in check. In people with kidney problems, though, sodium and bicarbonate can stick around too long and cause trouble.
Practical Advice—Safe Use Starts With Honest Labels
From a journalist’s corner and as someone who grew up watching folks grab baking soda for everything, I’ve learned facts matter. The best safeguard isn’t hiding sodium bicarbonate in the back of the cupboard. It's reading ingredient lists and paying attention to how much goes in—both in medicine and that batch of biscuits.
Doctors and pharmacists aren’t just for emergencies. They’ve got advice worth listening to, especially if someone already takes prescription meds or battles health conditions. There’s no shortcut for honest conversations about side effects—old-fashioned questioning and not just a quick search online.
Managing sodium intake works better as a habit, not an afterthought. This means choosing foods and remedies wisely, measuring doses, and skipping unnecessary antacid routines if heartburn pops up often. Instead of masking the pain, it’s smarter to ask what keeps bringing it back. This keeps side effects from creeping in and lets sodium bicarbonate remain the helpful, humble tool it’s always been—used right, not overdone.
The Humble Power of Baking Soda
It sits on most kitchen shelves, quietly waiting to deodorize a fridge or fluff up a batch of pancakes. Sodium bicarbonate, better known as baking soda, earns its spot as a true household staple. But every time I grab that old box wedged behind the flour and sugar, I can’t help but notice the little things that keep it at its best. This isn’t just about not letting it spill; it’s got everything to do with how we handle a product we trust to cook, clean, and sometimes even help with heartburn.
Moisture: Baking Soda’s Biggest Enemy
Let’s get real: sodium bicarbonate loves soaking up moisture. Open up a forgotten tub and you’ll find caked powder that barely sifts. Humid air turns good baking soda into a useless lump because it reacts with moisture and loses its punch. In my own place, I’ve watched a box kept too close to the stove turn into a solid mass. That lesson taught me to keep new boxes in a tight-lidded jar, which works anywhere—kitchen, bathroom, or even under the sink.
Why Light and Heat Matter
Pantries can get warm, especially with ovens cranking in the summer. I try to keep the jar in a cabinet away from direct sunlight and heat. Sodium bicarbonate breaks down over time if left in hot or brightly lit places. There’s an obvious chemistry here: heat and light mess with the fine balance that lets baking soda do its thing. If you stash your box next to the stove or in a sun-soaked window, say goodbye to those perfect cookies and bubbling science-project volcanoes.
Odors Sneak In, Too
Baking soda doesn’t just mask odors in the fridge—it absorbs them. Keep an open box by onions or dish soap, and everything you bake starts tasting a bit off. I learned this the hard way after storing soda in a loose bag above a garbage can. Tossing out a batch of weird-tasting muffins convinced me that a sealed container is the only way to go. It also stops curious pets or kids from knocking everything over and creating a powdery mess.
Shelf Life and Household Wisdom
Sodium bicarbonate won’t keep forever. Over time, even in a sealed container, it starts to lose its effectiveness. Most brands suggest a shelf life printed on the box—usually around two years. Simple home tests can help: drop a pinch into vinegar, and if it bubbles fiercely, it’s still potent. No fizz? Toss it or save it for cleaning. It’s a trick my parents swore by and one that saved many a recipe, especially during the holidays.
Solutions Start at Home
Storing sodium bicarbonate right isn’t complicated, but doing it wrong wastes money and ruins more than a few meals. All it takes is a dry, cool spot and a container with a tight seal. In my experience, glass jars with rubber gaskets work best—and they look good on a shelf. A little attention to storage turns a common white powder into the multi-tasking helper it’s meant to be.
Value in the Details
No tricked-out storage system required, just some respect for the basics. Look after your baking soda and it always does the job: keeping food tasty, fridges fresh, and homes running clean. That small gesture—putting the box in a sealed jar—might not feel like much, but it makes every bit of difference.